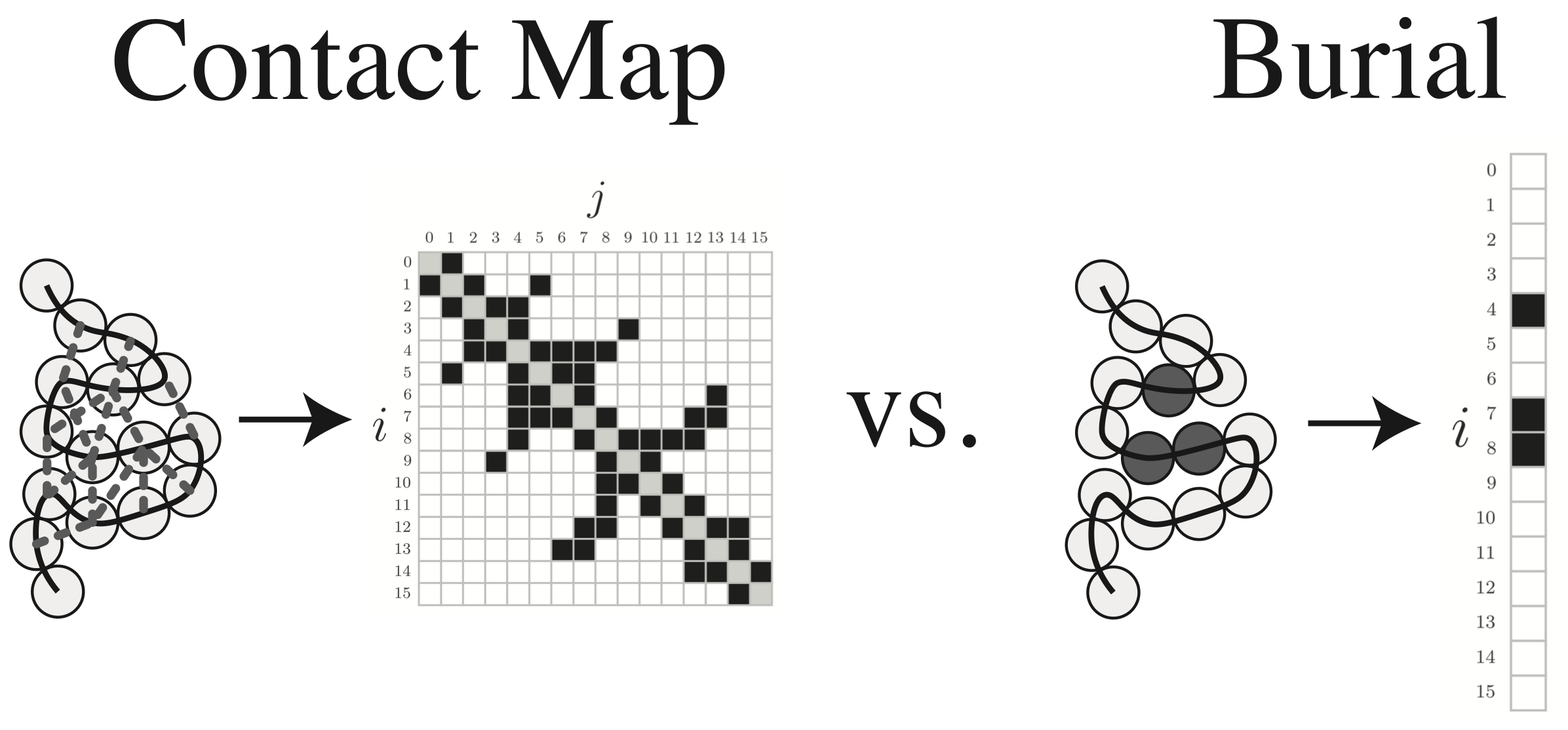
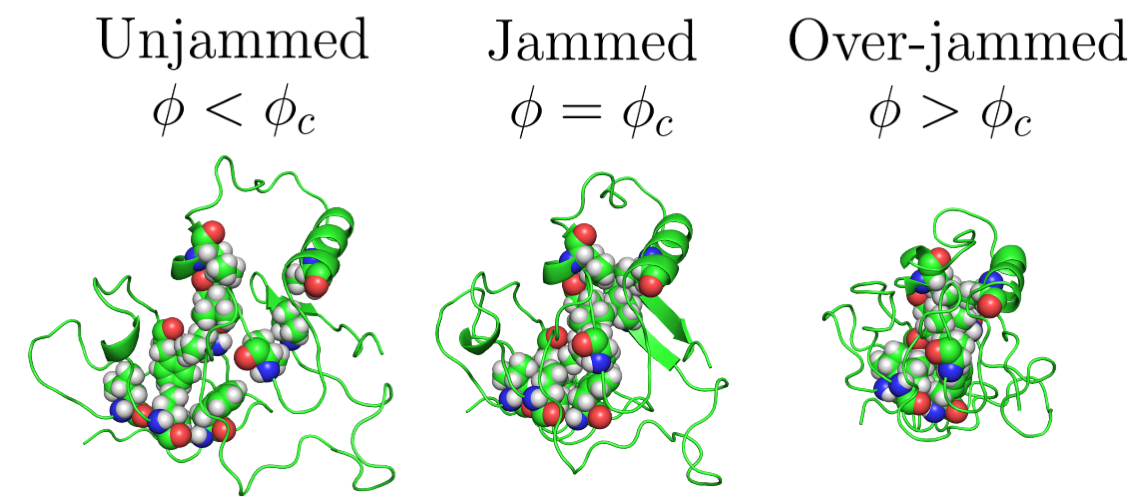
About
I'm a computational biophysicist studying how proteins fold and how tissues flow. I received my Ph.D. in computational biology and bioinformatics from Yale University in 2024, advised by Corey O'Hern. My doctoral work was equal parts protein structural informatics and soft matter physics. Since 2024, I have been a postdoctoral researcher in the Syracuse Physics Department, working with M. Lisa Manning. In collaboration with Alessandro Mongera, we have developed new discrete element models for dynamic and sparse tissues under tension. See below for highlights of my work and my CV.